Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Muramatsu, Yasuji; Kaneyoshi, Takahiro*; Gullikson, E. M.*; Perera, R. C. C.*
Spectrochimica Acta, Part A, 59(9), p.1951 - 1957, 2003/07
Angle-resolved soft X-ray emission and absorption spectra in the BK and NK regions of hexagonal BN were measured using polarized synchrotron radiation. The take-off/incident-angle-dependence on the spectral features in both X-ray emission and absorption is clearly observed. The configuration of the s and p orbitals, which were calculated using discrete variaional (DV) -Xa molecular orbital calculations, explains the angle-resolved soft X-ray emission and absorption spectra. The relative peak intensity of the 394-eV peak in the NK X-ray emission provides useful information about the BN layer ordering.
Sasaki, Teikichi; Chugan, Nobuhiko*; Muramatsu, Yasuji*
Nuclear Instruments and Methods in Physics Research A, 467-468(Part.2), p.1489 - 1492, 2001/07
Times Cited Count:3 Percentile:28.27(Instruments & Instrumentation)no abstracts in English
Muramatsu, Yasuji; Hirono, Shigeru*; Umemura, Shigeru*; Ueno, Yuko*; Hayashi, Takayoshi*; Grush, M. M.*; Gullikson, E. M.*; Perera, R. C. C.*
Carbon, 39(9), p.1403 - 1407, 2001/06
Times Cited Count:18 Percentile:58.33(Chemistry, Physical)no abstracts in English
Muramatsu, Yasuji; Ueno, Yuko*; Ishiwata, Yoichi*; Eguchi, Ritsuko*; Watanabe, Masamitsu*; Shin, S.*; Perera, R. C. C.*
Carbon, 39(9), p.1359 - 1402, 2001/06
no abstracts in English
 single-crystal
single-crystalAgui, Akane; Guo, J.-H.*; Sathe, C.*; Nordgren, J.*; Hidaka, M.*; Yamada, I.*
Solid State Communications, 118(12), p.619 - 622, 2001/06
Times Cited Count:5 Percentile:32.77(Physics, Condensed Matter)no abstracts in English
Muramatsu, Yasuji; Takenaka, Hisataka*; Ueno, Yuko*; Gullikson, E. M.*; Perera, R. C. C.*
Applied Physics Letters, 77(17), p.2653 - 2655, 2000/10
Times Cited Count:13 Percentile:51.49(Physics, Applied)no abstracts in English
Agui, Akane; Sathe, C.*; Guo, J.-H.*; Nordgren, J.*; Mankefors, S.*; Nilsson, P. O.*; Kanski, J.*; Andersson, T. G.*; Karlsson, K.*
Applied Surface Science, 166(1-4), p.309 - 312, 2000/10
Times Cited Count:2 Percentile:15.67(Chemistry, Physical)no abstracts in English
Terauchi, Masami*; Takahashi, Hideyuki*; Murano, Takanori*; Imazono, Takashi; Koike, Masato; Nagano, Tetsuya*; Koeda, Masaru*
no journal, ,
The chemical bonding state of titanium dioxide is clearly revealed by using in combination with soft X-ray grating spectrometer and SEM. In a simple ionic model considered as Ti O
O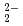 in TiO
in TiO , Ti
, Ti ion has no 3
ion has no 3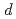 electron, but Ti
electron, but Ti 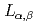 emission spectra resulting from the electron transition from 3
emission spectra resulting from the electron transition from 3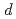 valance band to the
valance band to the  core holes can be observed. Thus, it indicates that the
core holes can be observed. Thus, it indicates that the 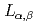 emissions of Ti originates from the covalent bonding between Ti and O atoms. The peak position of the
emissions of Ti originates from the covalent bonding between Ti and O atoms. The peak position of the 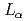 band of TiO
band of TiO , brookite, is found to be placed on the lower energy side than that of metal-Ti, but it is opposite to the theoretical prediction. This means that the
, brookite, is found to be placed on the lower energy side than that of metal-Ti, but it is opposite to the theoretical prediction. This means that the 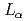 intensity profile is not suitable for probing chemical shift due to the influence from the density of states of the valence band. On the other hand, the peak shift of the
intensity profile is not suitable for probing chemical shift due to the influence from the density of states of the valence band. On the other hand, the peak shift of the 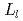 band caused by transitions between inner-shell levels is on the higher energy side than that of metal-Ti. It is found to be reasonable for the cation chemical shift.
band caused by transitions between inner-shell levels is on the higher energy side than that of metal-Ti. It is found to be reasonable for the cation chemical shift.
Terauchi, Masami*; Takahashi, Hideyuki*; Murano, Takanori*; Koike, Masato; Imazono, Takashi; Koeda, Masaru*; Nagano, Tetsuya*
no journal, ,
The 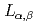 emission spectra of Ti resulting from the electron transition from
emission spectra of Ti resulting from the electron transition from 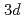 valance band to the
valance band to the  core holes were observed. These reflect the chemical bonding states for the covalent bonding between Ti and O atoms. The peak position of the
core holes were observed. These reflect the chemical bonding states for the covalent bonding between Ti and O atoms. The peak position of the 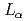 band is found to be placed on the lower energy side than that of metal-Ti, but which is opposite to the theoretical prediction. This means that the
band is found to be placed on the lower energy side than that of metal-Ti, but which is opposite to the theoretical prediction. This means that the 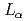 intensity profile is not suitable for probing the chemical shift due to the influence from the density of states of the valence band. On the contrary, the peak shift of the
intensity profile is not suitable for probing the chemical shift due to the influence from the density of states of the valence band. On the contrary, the peak shift of the 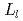 band caused by transitions between inner-shell levels is on the higher energy side. It is found to be reasonable for the cation chemical shift.
band caused by transitions between inner-shell levels is on the higher energy side. It is found to be reasonable for the cation chemical shift.
Koike, Masato
no journal, ,
Electron microscope and has a nearly 90-year history, has a lot of contributions to the observation and analysis of the micro-region. Therefore elemental analysis, structural analysis, energy dispersive spectrometer for the purpose of state analysis (EDS), wavelength dispersive spectrometer (WDS) and many of the spectrometer have been developed. However, a practical electron microscope spectrometer in the soft X-ray region required for the analysis of light elements such as lithium was present. To overcome this problem, the group of Tohoku University, JEOL Ltd., Shimadzu Corporation, and Japan Atomic Energy Agency have developed a soft X-ray spectrometer for electron microscopy to achieve high energy resolution by combining varied-line-spacing aberration-corrected diffraction gratings and a high sensitivity X-ray CCD camera. This instrument allows us parallel detection like EDS, and it is possible to perform high energy resolution analysis, e.g., 0.3 eV (Fermi edge Al-L), in a wide energy region of 50 eV  4 keV. By spectroscopic system design of the new development, spectra including various energies can be measured simultaneously without moving diffraction grating and detector. Moreover, taking advantage of a high energy resolution, it is possible to collect the state analysis map. We describe the development of flat-field varied-line-spacing holographic laminar type diffraction grating, which is the heart of this device, and give an overview of the spectrometer system as well as an summary of measured data including mapping.
4 keV. By spectroscopic system design of the new development, spectra including various energies can be measured simultaneously without moving diffraction grating and detector. Moreover, taking advantage of a high energy resolution, it is possible to collect the state analysis map. We describe the development of flat-field varied-line-spacing holographic laminar type diffraction grating, which is the heart of this device, and give an overview of the spectrometer system as well as an summary of measured data including mapping.